Ali Z. Qandoos1, Hanan A. Ahmed2, and Wafaa A. Abd El-Ghany1*
1 Faculty of Veterinary Medicine, Cairo University, Giza, Egypt
2 Central Laboratory for Evaluation of Veterinary Biologics, Cairo, Egypt
* Corresponding author: Wafaa A. Abd El-Ghany, Faculty of Veterinary Medicine, Cairo University, Giza, Egypt. Email: wafaa.ghany@yahoo.com
ABSTRACT
The current study aimed to characterize Pasteurella multocida (P. multocida) isolates from layers and breeder chickens in Egypt regarding in-vitro antibiotic sensitivity and resistance pattern. In doing so, spleen, liver, lungs, and heart, were taken aseptically from chickens suffering from drop in egg production, septicemia, respiratory manifestations, and mortalities between 2016 and 2017. To isolate bacteria, samples were grown on a modified Das medium. Moreover, microscopic appearance and biochemical characteristics were used to identify pure colonies of P. multocida isolates. In the next step, in-vitro antibiotic sensitivity testing was performed on the isolated P. multocida. The findings indicated that P. multocida was found in 36 isolates out of 330 investigated chicken flocks. Small glistering, mucoid, grayish, and dew drop P. multocida colonies were discovered during the culture analysis. Pasteurella multocida isolates were Gram-negative coccobacilli using the microscope. Catalase, indole generation, H2S production, nitrate reduction, and oxidase tests were all positive for the sample, however, methyl red, urease activity, Voge’s proskaur, and gelatin liquefaction tests were all negative. They also fermented glucose, mannose, fructose, sucrose, mannitol, xylose, and sorbitol without producing gas but not lactose, arabinose, maltose, inositol, salicin, raffinose, or dulcitol. Isolated P. multocida strains were sensitive to tetracycline, erythromycin, trimethoprim/sulphamethoxazole, norfloxacin, ofloxacin, penicillin, chloramphenicol, and azithromycin, while resistant to ampicillin and clindamycin. Cefoperazone, gentamycin, and streptomycin all showed intermediate sensitivity.
1. Introduction
Fowl cholera (FC) is a contagious disease caused by Gram-negative bacteria, Pasteurella multocida (P. multocida). This disease remains a significant obstacle for poultry production in many countries in the world as it causes severe economic losses for domestic and backyard birds1,2. Fowl cholera takes different infection forms, including peracute, acute with high mortalities and morbidities, and chronic localized ones3. The bacterium, P. multocida, is usually present in the upper respiratory tract, pharynx, and cloacae of birds. Thus, isolation and identification of the organism from clinical samples are very important for the diagnosis of the disease. Vaccines are used against FC, but the infection remains in poultry flocks.
Antimicrobials resistance of bacteria has become a great problem in human and veterinary medicine4. Different antimicrobials have been widely used for the treatment of P. multocida with varying results depending on the species, time, geographical origin, and the type of the used drug5,6. Strains of P. multocida are susceptible to most of the widely used commercial antimicrobial agents. However, haphazard, indiscreet, and prolonged usage of antimicrobials for the treatment of P. multocida accelerate the emergence of multidrug resistance to commonly used chemotherapeutic agents7. The antibiotic resistance increases the incidence of P. multocida infection and subsequently affects the economy of the locality.
Therefore, the aim of this study was to characterize P. multocida isolates from Egyptian layer and breeder chicken flocks and determine the in vitro antibiotic sensitivity of isolates to different antimicrobial agents.
2. Materials ad Methods
2.1. Bacteriology
Samples were collected from layers and breeder chicken flocks inEl-Sharqia, El-Gharbia, El-Qalubia, and El-Minofia governorates, Egypt during the period from 2016 to 2017. The flocks were suffered from respiratory manifestations, septicemia, drop in egg production, and mortalities. The samples of liver, heart, spleen, and lungs were collected from freshly dead birds, inoculated in brain heart broth, and incubated at 37° C for 18-24 hrs. Subsequent selective sub-culturing of P. multocida isolates was done on modified Das media under aerobic conditions at 37°C for 48 hours to obtain pure cultures8. Gram staining was used for morphological identification of colonies9. Biochemical identification was made according to Quinn et al.10.
2.2. In-vitro antibiotic sensitivity test
Isolated strains of P. multocida were tested for their susceptibility to 13 antimicrobial agents obtained from Oxoid Laboratories, UK The antibiotic discs were norfloxacin (NOR, 10 µg), gentamycin (CN, 10 µg), tetracycline (TE, 30 µg), erythromycin (E, 15 µg), streptomycin (S, 10 µg), cefoperazone (CEP, 75 µg), trimethoprim/sulphamethoxazole (SXT, 1.25/23.75 µg), ampicillin (AM, 10 µg), ofloxacin (OFX, 5 µg), chloramphenicol (C, 30 µg), penicillin G (P, 10 µg), azithromycin (AZM, 15 µg), and clindamycin (DA, 2 µg). Pure P. multocida colonies were picked and suspended in sterile saline and the turbidity was adjusted to 0.5 Mcfarland standard tube. The sterile cotton swab was dipped into the prepared inoculum tube, spread uniformly into Muller Hinton agar. The antibiotic discs were dispensed on the surface of the agar using forceps and the plates were incubated at 37oC for 24 hours. The zones of inhibition were measured and recorded to determine the sensitivity or resistance of P. multocida to the tested drug
according to the standardized protocol by the Clinical and Laboratory Standards Institute11.
3. Results and discussion
Pasteurella multocida is the cause of avian cholera, a disease that has been described worldwide and causes great losses to the poultry industry12. Healthy carriers and chronic forms of the infection were well described13. Antimicrobial treatments have been extensively used for P. multocida with varying success5.
Isolation of P. multocida on DAS media showed small glistering, grayish, mucoid, and dew drop colonies. Gram-negative coccobacilli were observed in stained smears from suspected P. multocida colonies. Suspected P. multocida isolates were positive for catalase, oxidase, indole production, nitrate reduction, and H2S production tests, while negative for methyl red, Voges-Proskauer, urease activity, and gelatin liquefaction tests. Moreover, they fermented glucose, fructose, mannose, mannitol, sucrose, sorbitol, and xylose without gas production but not arabinose, inositol, lactose, maltose, salicin, dulcitol, and raffinose. These findings are in accordance with Kawamota14, Arora et al.7, Purushothaman et al.15, and Balasubramanium and Gopalakrishnamurthy16. Isolation of P. multocida from the liver of chickens was recorded17.
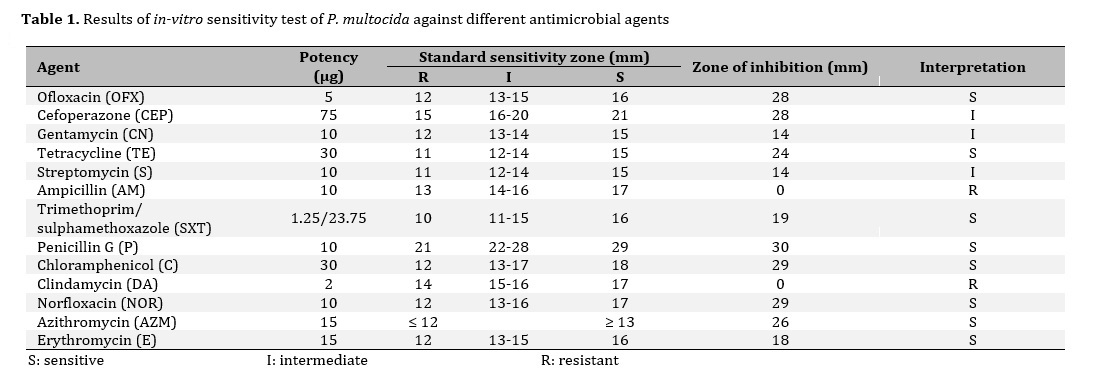
The sensitivity of P. multocida to different antibiotics is shown in Table (1) and Figure (1). In the present study, the result of in-vitro antibiotic sensitivity test indicated that P. multocida was sensitive to ofloxacin, tetracycline, trimethoprim/sulphamethoxazole, penicillin, chloramphenicol, norfloxacin, azithromycin, and erythromycin, while resistant to ampicillin and clindamycin. Intermediate sensitivity was observed for cefoperazone, gentamycin, and streptomycin.
Sarangi and Panda18 studied the antibiotic sensitivity test of P. multocida isolates and found that the organisms were sensitive to enrofloxacin, gentamycin, levofloxacin, gatifloxacin, and chloramphenicol, but resistant to penicillin G, streptomycin, sulfadiazine, cephalexin, cephotaxim, and ampicillin. Similar sensitivity was recorded by Hirsh et al.19 and Shivachandra et al.20 who demonstrated the susceptibility of P. multocida to chloramphenicol, enrofloxacin, gentamycin, tetracycline, penicillin G., streptomycin, sulphonamides, and trimethoprim.