Alireza Sadeghi1*, Ali Shabestari Asl2, Daryoush Babazadeh3, and Pouria Ahmadi Simab4
1 Doctor of Veterinary Medicine, Faculty of Veterinary Medicine, Tabriz Branch, Islamic Azad University, Tabriz, Iran
2 Department of Clinical Science, Faculty of Veterinary Medicine, Tabriz Branch, Islamic Azad University, Tabriz, Iran
3 School of Veterinary Medicine, Shiraz University, Shiraz, Iran
4 Faculty of Veterinary Medicine, Sanandaj Branch, Islamic Azad University, Sanandaj, Iran
* Corresponding author: Alireza Sadeghi, Faculty of Veterinary Medicine, Tabriz Branch, Islamic Azad University, Tabriz, Iran
Comparison of Three Different Glucose-lowering drugs on Serum Levels of Glucose and Pancreas Histopathology in Streptozotocin-Induced Diabetic Rats
A B S T R A C T
Introduction: Diabetes mellitus is a metabolic disorder resulting from a defect in insulin secretion, insulin action, or both. The aim of the present study was to compare the effect of three different blood glucose-lowering drugs in streptozotocin-induced diabetic rats.
Materials and methods: 60 male Wistar rats (220–250 g and 2-3 months of age) were selected for the current study, and they then were divided into five equal groups. Group 1 included healthy control rats receiving standard diet, group 2 involved diabetic rats receiving standard diet plus acarbose (25mg/kg/day) via gastric feeding tube daily for 8 weeks, group 3 embraced diabetic rats receiving standard diet plus pioglitazone (1 mg/kg/day) via gastric feeding tube daily for 4 weeks, and group 4 received of diabetic rats receiving standard diet plus repaglinide (10 mg/kg/day) via gastric feeding tube daily for 4 weeks. Diabetes was induced by intraperitoneal injection of streptozotocin at a dosage of 65 mg/kg body weight. At the end of the study, the samples were taken for histopathological investigation of pancreas and serum glucose levels. The mean diameter of pancreatic islets and the percentage of beta and alpha cells were calculated in all groups.
Results: The fasting blood glucose in three treated and normal control rats was significantly less than diabetic control group. One hour after treatment the blood glucose level reduced significantly in three treated and normal control rats compared to diabetic control group. On day 7, the percentage of alpha cells in the pioglitazone and acarbose groups increased significantly compared to the diabetic control group. On day 28, the percentage of beta cells in the treated groups increased significantly compared to normal and diabetic control groups. On day 28, the mean of islet diameter in the treated groups increased significantly compared to the normal and diabetic control groups. On day 28, the percentage of alpha cells in the repaglinide group was significantly reduced compared to the diabetic control group.
Conclusion: Among the administrated drugs, the pioglitazone had the most positive effects on controlling blood glucose, increasing beta cells as well as improving the diameter of pancreatic islets.
Key words: Acarbose, Diabetes mellitus, Pioglitazone, Repaglinide, Streptozotocin
1. Introduction
Diabetes mellitus is one of the most common endocrine disorders affecting almost 6% of the world’s population. According to the report of the International Diabetes Federation in 2001, the number of diabetic patients will reach 300 million in 2025. More than 97% of these patients will have type II diabetes1. Diabetes mellitus is characterized by hyperglycemia and is associated with disturbances in carbohydrate, protein and fat metabolism which occurs secondary to an absolute (type І) or relative (type ІІ) lack of insulin2.
Acarbose is an alpha-glycosidase inhibitor and antidiabetics are used for the treatment of diabetes. Acarbose shows its effect by inhibiting intestinal enzymes (alpha-glycosidases), thereby interfering with the catabolism of disaccharides, oligosaccharides, and polysaccharides in the intestines. Thus, digestion of carbohydrates is delayed depending on the dose, and more importantly, glucose release and its presence in the blood slows down. Moreover, both fluctuations in daily blood sugar and average blood sugar level decrease as a result of this delayed glucose intake through the intestines
with acarbose. Acarbose also reduces abnormal high concentrations of glycosylated hemoglobin3-5. A possible explanation for the discrepancies is an observation that acarbose is significantly more effective in patients eating a relatively high carbohydrate Eastern diet6,7. Acarbose inhibits enzymes (glycoside hydrolases) are required
to digest carbohydrates, specifically, alpha-glucosidase enzymes in the brush border of the small intestines and pancreatic alpha-amylase. Pancreatic alpha-amylase hydrolyzes complex starches to oligosaccharides in the lumen of the small intestine, whereas the
membrane-bound intestinal alpha-glucosidase hydrolyzes oligosaccharides, trisaccharide’s, and disaccharides to glucose and other monosaccharides in the small intestine. Inhibition of these enzyme systems reduces the digestion rate of complex carbohydrates. Less glucose is absorbed because the carbohydrates are not broken down into glucose molecules. In diabetic patients, the short-term effect of these drugs is a decrease in current blood glucose levels and the long-term effect is a reduction in Hemoglobin A1c (HbA1c) level8. Acarbose reduces postprandial plasma glucose and may improve metabolic control in non-insulin-dependent diabetes mellitus when combined with the diet9. This reduction averages an absolute decrease of 0.7%, which is a decrease of about 10% in typical HbA1c values in diabetes studies6.
Pioglitazone, a thiazolidinedione (TZD) insulin sensitizer, is a peroxisome proliferator-activated receptor gamma (PPAR-γ) agonist. It increases insulin sensitivity by regulating the expression of a variety of genes involved in carbohydrate and lipid metabolism, increases Glucose transporter-4 (GLUT-4) and glucokinase activity, decreases phosphoenolpyruvate carboxykinase (PEPCK) expression, and decreases production by a fat cell of several mediators that may cause insulin resistance, such as tumor necrosis factor α (TNF α) and resistin10,11. As a result, pioglitazone reduces insulin resistance in the liver and peripheral tissues, increases the expense of insulin-dependent glucose, decreases withdrawal of glucose from the liver, and reduces the quantity of glucose, insulin, and glycated hemoglobin in the bloodstream. Regardless of being clinically insignificant, pioglitazone decreases the level of triglycerides and increases that of High-density lipoproteins (HDL) without changing Low-density lipoproteins (LDL) and total cholesterol in patients with disorders of lipid metabolism although statins are the choice of drug for this issue. More recently, pioglitazone and other active TZDs have been shown to bind to the outer mitochondrial membrane protein mitoNEET with an affinity comparable to that of pioglitazone for PPARγ12,13. Pioglitazone increases hepatic and peripheral insulin sensitivity, thereby inhibiting gluconeogenesis and increasing peripheral and splanchnic glucose uptake14.
Repaglinide is an anti-diabetic drug in the class of medications known as meglitinides, and was invented in 1983. Repaglinide lowers blood glucose by stimulating the release of insulin from the pancreas by closing ATP-dependent potassium channels in the membrane of the beta cells. This depolarizes the beta cells, opens the calcium channels of cells, and the resulting calcium influx induces insulin secretion15. Repaglinide is metabolized by cytochrome CYP3A4 in the liver16. The aim of the present study was to compare the serum level of glucose and histopathological effects of three different blood glucose-lowering drugs in streptozotocin-induced diabetic rats.
2. Materials and Methods
2.1. Ethical approval
All procedures were approved by the Animal Care Committee of Veterinary Medicine, Islamic Azad University, Tabriz Branch, Iran. The principles of laboratory animal care were followed, and specific international laws were observed.
2.2. Animals
60 male Wistar rats weighing approximately 220–250 g with 2-3 months of age were acclimated to laboratory conditions for 4 weeks, followed by maintenance under controlled temperature (25-28°C) and light (12/12-hours light/dark cycle) conditions. Animals received standard extruded pellet and water ad libitum until treatment. Rats selected for the study were purchased from Animal House, Islamic Azad University, Iran, and randomly divided into five equal groups. Group 1 included healthy control rats who received a standard diet, group 2 diabetic rats received standard diet plus acarbose (25mg/kg/day) via gastric feeding tube daily for 4 weeks, group 3 involved diabetic rats received standard diet plus pioglitazone (1mg/kg/day) via gastric feeding tube daily for 4 weeks, and group 4 entailed diabetic rats who received standard diet plus repaglinide (10 mg/kg/day) via gastric feeding tube daily for 4 weeks.
2.3. Diabetes infusion
Diabetes was induced by intraperitoneal injection of streptozotocin (single dose, Sigma, St. Louis, Mo, USA) at a dosage of 65 mg/kg body weight. The STZ was extemporaneously dissolved in 0.1 M cold sodium citrate buffer, pH 4.5. After 48 hours, animals with fasting blood glucose higher than 250 mg/dl were considered diabetic and were used in the present study17.
2.4. Micrometric perusal methods
The pancreases fixed in a 10% neutral-buffered formalin solution were embedded in paraffin and used for histopathological examination. Therefore, 5 micrometer thick sections were cut, deparaffinized, hydrated, and stained with hematoxylin-eosin. A minimum of 10 fields for each slide was examined and assigned for the severity of changes using scores on a scale of mild (1+), moderate (2+), and severe (3+) damage18-21.
2.5. Blood sample
Fasting blood glucose was estimated by using the Bio check Glucose Test Strip (Accu-chek sensor) of Roche Diagnostics, Germany at the end of study (6 six rats in each group). The animals of different groups were sacrificed under light anesthesia (diethyl ether) at the end of the treatment (6 six rats in each group).
2.6. Statistical analysis
The Statistical Package for Social Sciences (SPSS Inc., Chicago, IL, USA), version 13, was used for statistical analysis. All data are presented as mean ± SEM. Before statistical analysis, all variables were checked for normality and homogeneity of variance using the Kolmogorov-Smirnoff and Levene tests, respectively. The data obtained were tested by ANOVA followed by Tukey’s posthoc multiple comparison test. The p value less than 0.05 was considered statistically significant.
3. Results
3.1. Fasting blood glucose levels
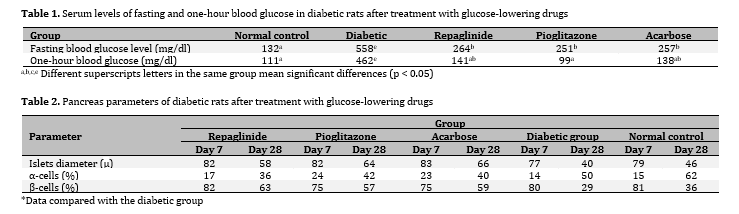
Mortality was not seen in this study. Three treated and diabetic control groups showed significant differences with normal control group (p < 0.05). The three treated and normal control rats indicated significant differences with diabetic control group (p < 0.05).
3.2. Blood glucose levels one hour after the last treatment
The three treated and normal control groups showed a significant difference from the diabetic control group
(p < 0/05, Table 1). It should be noted that the best effect on one-hour glucose was in the pioglitazone group, which was significantly different from the diabetic control group (p < 0/05) however it did not have a significant difference compared to other treatment groups (p > 0/05, Table 1).
3.2. Histopathological findings
On day 7, the percentage of alpha cells in the pioglitazone and acarbose groups increased significantly compared to the diabetic control group (p < 0.05, Table 2). On day 28, the percentage of beta cells in the treated groups increased significantly compared to normal and diabetic control groups (p < 0.05, Table 2). On day 28, the mean of islet diameter in the treated groups increased significantly compared to the normal and diabetic control groups
(p < 0.05, Table 2). On day 28, the percentage of alpha cells in the repaglinide group was significantly reduced compared to the diabetic control group (p < 0.05, Table 2). The obtained results indicated that anti-diabetic drugs have beneficial effects on regeneration of pancreatic islets and cells which were so obvious in beta cells (Table 2).